Ionic EquilibriumHard
Question
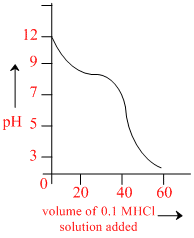
When weak base solution (50 ml of 0.1 N NH4OH) is titrated with strong acid 0.1 N HCl, the pH of solution initially decreases fast and then decreases slowly till near equivalence point (as shown) in the figure. Which of the following is true.

Options
A.The slow decrease of pH is due to formation of an acidic buffer solution after addition of some HCl
B.The slope of shown pH graph will be minimum when 25 ml of 0.1 N HCl is added
C.The slow decrease of pH is due to formation of a basic buffer solution
D.The initial fast decrement in pH is due to fast consumption of free OH- ions by HCl.
Solution
Initial decrement is due to consumption of free OH– ions, then slow decrement in pH is due to basic buffer solution and minimum slope will be there when there is best buffer action ([salt] / [base] = 1)
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The strength of an aqueous solution of I2 can be determined by titrating the solution with standard solution of:...A 2.5 g impure sample containing weak monoacidic base (Molecular weight = 45) is dissolved in 100 ml water and titrated ...If pH of a saturated solution of Ba(OH)2 is 12, the value of its Ksp is...Number of H+ ions present in 10 mL of solution of pH = 3 are:...The vapour pressure of a given liquid will decrease if....