Ionic EquilibriumHard
Question
Which statement/relationship is correct?
Options
A.pH of 0.1 M HNO3, 0.1M HCl, 0.1M HI are not equal.
B.pH = - log 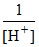
C.At 25oC the pH of pure water is 7.
D.The value of pKw at 25oC is 7.
More Ionic Equilibrium Questions
The solubility product of AgCl is 1.0 × 10–10. The equilibrium constant of the reactionAgCl(s) + Br–$\rightleftharpoons$...What is the pH of 10−7 M-HCl solution at 25o C?...10 mL, of a strong acid solution of pH = 2.000 are mixed with 990 mL of another strong acid solution of pH = 4.000. The ...The pH at the equivalence point when a solution of 0.01 M-CH3COOH is titrated with a solution of 0.01 M-NaOH, is (pKa of...If pKb for fluoride ion at 25°C is 10.3, the ionization constant of hydrofluoric acid in water at this temperature is (l...