Ionic EquilibriumHard
Question
10 mL, of a strong acid solution of pH = 2.000 are mixed with 990 mL of another strong acid solution of pH = 4.000. The pH of the resulting solution will be:
Options
A.4.002
B.4.000
C.4.200
D.3.7
Solution
[H+] after mixing = 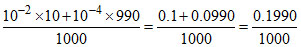 = 1.99 × 10-4.
= 1.99 × 10-4.
∴ pH = 4 - 0.3 = 3.7
∴ pH = 4 - 0.3 = 3.7
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The pH of solution containing 0.10 M sodium acetate and 0.03 M acetic acid is(pKa for CH3COOH = 4.57)...Aniline behaves as a weak base. When 0.1 M, 50 ml solution of aniline was mixed with 0.1 M, 25 ml solution of HCl the pH...The buffer capacity (β) for a weak acid (A) – conjugate base (B) buffer is defined as the number of moles of strong acid...Among the following acids which has the lowest pKa value?...How many moles of NaOH must be removed from one litre of aqueous solution to change its pH from 12 to 11 ?...