Ionic EquilibriumHard
Question
Which of the following relations is correct ?
Options
A.ᐃGo = RT ln Keq
B.[H3O+] = 10pH
C.log 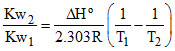
D.[OH-] = 10-7,
More Ionic Equilibrium Questions
Two buffer solutions, A and B, each made with acetic acid and sodium acetate differ in their pH by one unit, A has salt ...When 20 ml of 0.2 M-DCl solution is mixed with 80 ml of 0.1 M-NaOD solution, pH of the resulting solution becomes 13.6. ...To precipitate as Ag2S(s), all Ag+ present in 250 ml of a satrurated solution of AgBrO3 requires 3.36 cm3 of H2S(g) meas...A 50 ml solution of strong acid of pH = 1 is mixed with a 50 ml solution of strong acid of pH = 2. The pH of the mixture...Which of the following is acidic salt :-...