Ionic EquilibriumHard
Question
To precipitate as Ag2S(s), all Ag+ present in 250 ml of a satrurated solution of AgBrO3 requires 3.36 cm3 of H2S(g) measured at STP (Assuming H2S is enough soluble in water). Ksp of AgBrO3 is [Ksp(Ag2S) = 10-50, Ka(H2S) = 10-21]
Options
A.1.44 × 10-6
B.2.25 × 10-7
C.9 × 10-6
D.2.7 × 10-7
Solution
2Ag+ (aq) + H2S(g) → Ag2S (s) + 2H+ (aq) ; K = 1029
moles of H2S dissolved = 1.5 × 10-4
∴ moles of Ag+ precipitated = 3 × 10-4
∴ Ksp(AgBrO3) =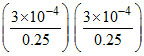 = 1.44 × 10-6
= 1.44 × 10-6
moles of H2S dissolved = 1.5 × 10-4
∴ moles of Ag+ precipitated = 3 × 10-4
∴ Ksp(AgBrO3) =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
How many moles of acetic acid should be added to 100 ml of 0.6 M formic acid solution such that the percentage dissociat...An amount of 0.16 g of N2H4 is dissolved in water and the total volume is made up to 500 ml. What is the percentage of N...20 ml, 5 M NaNO2 solution is mixed with 100 ml, 2M HNO2 solution to prepare a buffer solution of pH = 5.7. The Ka of HNO...At 25oC, the solubility product values of AgCl and AgCNS are 1.8 × 10-10 and 1.6 × 10-11 respectively. When a ...There exist an equilibrium between solid BaSO4, Ba2+ and SO42− ions in aqueous medium. Now, if equilibrium is disturbed ...