Ionic EquilibriumHard
Question
A 50 ml solution of strong acid of pH = 1 is mixed with a 50 ml solution of strong acid of pH = 2. The pH of the mixture will be nearly (log 5.5 = 0.74)
Options
A.0.74
B.1.26
C.1.76
D.1.5
Solution
[H+] = 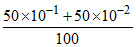 = 5.5 × 10-2 M.
= 5.5 × 10-2 M.
∴ pH = 2 - 0.74 = 1.26
∴ pH = 2 - 0.74 = 1.26
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A certain weak acid has a dissociation contant 1.0 × 10-4. The equilibrium constant for its reaction with a strong ...Solubility of BaF2 in a solution of Ba(NO3)2 will be represented by the concentration term:...At 30oC the solubility of Ag2CO3 (KSP = 8 × 10-12) would be greatest in one litre of:...After solid SrCO3 was equilibrated with a buffer at pH 8.6, the solution was found to have [Sr2+] = 2.0 × 10−4 M, what i...At infinite dilution the percentage dissociation of both weak acid and weak base is:...