Ionic EquilibriumHard
Question
Which of the following expression is not true ?
Options
A.[H+] = [OH-] = 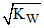 for a neutral solution at all temperatures.
for a neutral solution at all temperatures.
B.[H+] > 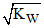 & [OH-] <
& [OH-] < 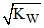 for an acidic solution
for an acidic solution
C.[H+] <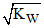 & [OH-] >
& [OH-] >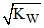 for an alkaline solution
for an alkaline solution
D.[H+] = [OH-] = 10-7 M for a neutral solution at all temperatures .
More Ionic Equilibrium Questions
The dissociation constant of a weak acid HX is 10–5. The buffer HX + NaX can be best used to maintain the pH in the rang...In a sautrated solution of the sparingly soluble strong electrolyte AgIO3(Molecular mass = 283) the equilibrium which se...A certain buffer solution contains equal concentration of X- and HX. The Kb for X- is 10-10. The pH of the buffer is :...Which of the following ids the strongest acid ?...A certain weak acid has a dissociation constant of 1.0 × 10-4. The equilibrium constant for its reaction with a str...