Ionic EquilibriumHard
Question
In a sautrated solution of the sparingly soluble strong electrolyte AgIO3(Molecular mass = 283) the equilibrium which sets in is
AglO3(6) ⇋ Ag+(aq) + IO-3(aq)
If the solubility product constant Ksp of AgIO3 at a given temperature is 1.0 × 10-8, what is the mass of AgIO3 contained in 100 ml of its saturated solution?
AglO3(6) ⇋ Ag+(aq) + IO-3(aq)
If the solubility product constant Ksp of AgIO3 at a given temperature is 1.0 × 10-8, what is the mass of AgIO3 contained in 100 ml of its saturated solution?
Options
A.28.3 × 10-2 g
B.2.83 × 10-3 g
C.1.0 × 10-7 g
D.1.0 × 10-4 g
Solution
AgIO3(s) ⇋ Ag+(aq) + IO-3(aq)
Let the solubility of AgIO3 be s
Ksp = [Ag+][IO-3]
1.0 × 10-8 = s2
s = 10-4 mol/litre
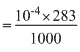 × 100
× 100
= 283 × 10-5
= 2.83 × 10-3 g/ 100 ml
Hence, (2) is correct.
Let the solubility of AgIO3 be s
Ksp = [Ag+][IO-3]
1.0 × 10-8 = s2
s = 10-4 mol/litre
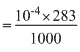 × 100
× 100= 283 × 10-5
= 2.83 × 10-3 g/ 100 ml
Hence, (2) is correct.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
M(OH)x has KSP 4 × 10-12 and solubility 10-4 M. The value of x is:...Calcium lactate is a salt of weak acid and represented as Ca(Lac)2. A saturated solution of Ca(Lac)2 contains 0.125 mole...A definite volume of a N/20 CH3COOH (pKa = 4.7447) is titrated with a strong base (NaOH). It is found that 80 equal size...The pH of an acidic buffer mixture is:...Which of the following solution will have pH close to 1.0 ?...