Ionic EquilibriumHard
Question
A certain weak acid has a dissociation constant of 1.0 × 10-4. The equilibrium constant for its reaction with a strong base is :
Options
A.1.0 × 10-4
B.1.0 × 10-10
C.1.0 × 1010
D.1.0 × 1014
Solution
HA OH- ⇋ A- + H2O
Weak acid + strong base
Keq =
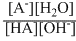
and [H2O] = Unity
∴ Keq =
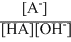 ..........(1)
..........(1)For dissociation of weak acid
HA ⇋ H+ + A-
Ka =
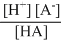 ..........(2)
..........(2) 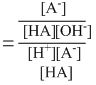
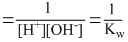
Keq = Ka / Kw
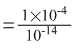
= 1.0 × 1010
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
For the identification of β - naphthol using dye test, it is necessary to use...Which one of the following statements is not true?...In which of the following solutions, the solubility of AgCN will be greater than that in pure water :Given Ksp(AgCN) = 4...The electrolytic reduction of nitrobenzene in strongly acidic medium produces :-...Which of the following is true...