Ionic EquilibriumHard
Question
A weak acid HA after treatment with 12 mL of 0.1 M strong base BOH has a pH of 5. At the end point the volume of same base required is 26.6 mL . Calculate Ka of acid is :
Options
A.1.8 × 10-5
B.8.22 × 10-6
C.1.8 × 10-6
D.8.2 × 10-5
Solution
For neutralisation :
Total Meq. of acid = Meq. of base = 26.6 × 0.1 = 2.66.
Now for partial neutralisation of acid.
HA + BOH BA + H2O
Meq. before reaction 2.66 1.2 0 0
Meq. after reaction 1.46 0 1.2 1.2
The resultant mixture acts as a buffer and [HA] and [BA] may be placed in terms of Meq. sincen volume of mixture is constant.
pH = - log Ka + log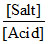
or 5 = - logKa + log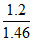 s
s
Ka = 8.22 × 10-6.
Total Meq. of acid = Meq. of base = 26.6 × 0.1 = 2.66.
Now for partial neutralisation of acid.
HA + BOH BA + H2O
Meq. before reaction 2.66 1.2 0 0
Meq. after reaction 1.46 0 1.2 1.2
The resultant mixture acts as a buffer and [HA] and [BA] may be placed in terms of Meq. sincen volume of mixture is constant.
pH = - log Ka + log
or 5 = - logKa + log
Ka = 8.22 × 10-6.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
What concentration of free CN– must be maintained in a solution that is 1.8 M-AgNO3 and 0.16 M-NaCl to prevent AgCl from...In which of the following cases, pH is greater than 7 ?...What is the ratio of pH of a solution containing 1 mole of CH3COONa and 1 mole of HCl per litre and the other solution c...A solution is prepared by dissolving 1.5 g of a monoacidic base into 1.5 kg of water at 300 K, which showed a depression...Complete hydrolysis of cellulose gives...