Ionic EquilibriumHard
Question
A solution is prepared by dissolving 1.5 g of a monoacidic base into 1.5 kg of water at 300 K, which showed a depression in freezing point by 0.165oC. When 0.496 g of the same base titrated, after dissolution, requires 40 mL of semimolar H2SO4 solution. If Kf of water is 1.86 K kg mol-1, then select the correct sattements (s) out of the following (assuming molarity = molality)
Options
A.The pH of the solution of weak base is 12.9.
B.The ionisation constant of the base is 8 × 10-3.
C.he osmotic pressure of the aqueous solution of base is 21.67 atm
D.The base is 10% ionized in aqueous solution.
Solution
Calculation of molecular weight of BOH:
Eq of base = Eq of H2SO4 used
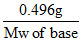 × 1 = 40 × 10-3 ×
× 1 = 40 × 10-3 × 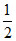 × 2(n factor)
× 2(n factor)
∴ Mw of BOH = 12.4 gmol-1
Now: ᐃfT = iKfm
0.165 = i × 1.86 ×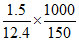 = i × 1.86 × 0.8 (So m = 0.8)
= i × 1.86 × 0.8 (So m = 0.8)
∴ i =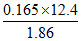 = 1.1
= 1.1
For weak base: 1 + α = 1.1, ⇒ α = 0.1 = 10% (Answer d)
Thus [OH-] = Cα = 0.8 × 0.1 = 8 × 10-2,
∴ pOH = 1.1, pH = 12.9 (Answer a)
So, Kb = Cα2 = 0.8 × (0.1)2 = 8 × 10-3, (Answer b)
Also; π = iMRT
= 1.1 × 0.8 × 0.0821 × 300
= 21.67 atm (Answer c)
Eq of base = Eq of H2SO4 used
∴ Mw of BOH = 12.4 gmol-1
Now: ᐃfT = iKfm
0.165 = i × 1.86 ×
∴ i =
For weak base: 1 + α = 1.1, ⇒ α = 0.1 = 10% (Answer d)
Thus [OH-] = Cα = 0.8 × 0.1 = 8 × 10-2,
∴ pOH = 1.1, pH = 12.9 (Answer a)
So, Kb = Cα2 = 0.8 × (0.1)2 = 8 × 10-3, (Answer b)
Also; π = iMRT
= 1.1 × 0.8 × 0.0821 × 300
= 21.67 atm (Answer c)
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which of the following solution will have a pH exactly equal to 8 ?...Liquid ammonia ionizes to slight extent. At −50°C, its self-ionization constant, K = [NH4+] [NH2−] = 10–30 M2. How many ...Some chemists at ISRO wished to prepare a saturated solution of a silver compound and they wanted it to have the highest...The acidic hydrolysis of ether (X) shown below is fastest when   Â...An amount of 2.0 M solution of Na2CO3 is boiled in a closed container with excess of CaF2. Very little amount of CaCO3 a...