Ionic EquilibriumHard
Question
If the equilibrium constant for the reaction of weak acid HA with strong base is 109, then pH of 0.1 M Na A is
Options
A.5
B.9
C.7
D.8
Solution
HA + NaOH → NaA + H2O; or HA + OH- → A- + H2O
Keq. = 109 =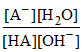 Also HA ⇋ H+ + A- Ka =
Also HA ⇋ H+ + A- Ka = 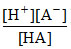
∴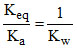 or Ka = 109 × 10-14 = 10-5 Thus for
or Ka = 109 × 10-14 = 10-5 Thus for
A- + H2O ⇋ HA + OH
[OH-] = Ch = C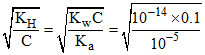 = 10-5 M
= 10-5 M
∴ [H+]= 10- and pH = 9
Keq. = 109 =
∴
A- + H2O ⇋ HA + OH
[OH-] = Ch = C
∴ [H+]= 10- and pH = 9
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Select CORRECT statement (s) -...The solubility product of AgCl is 10-10. The minimum volume (in L) of water required to dissolve 1.722 mg of AgCl is (mo...Which of the following is acidic salt :-...When 20 mL of M/20 NaOH is added to 10 mL of M/10 HCl, the resulting solution will...KSP if Mg(OH)2 is 1 × 10-12. 0.01M MgCI2 will show precipitation in a solution of pH greater than:...