NEETIonic EquilibriumHard
Question
The solubility product of AgCl is 10-10. The minimum volume (in L) of water required to dissolve 1.722 mg of AgCl is (molecular weight of AgCl = 143.5).
Options
A.10 lt.
B.2.2 lt.
C.1.2 lt.
D.20 lt.
Solution
Solubilility of AgCl in water = 10-5 mol/lt.
so volume of water required =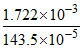 = 1.2 lt.
= 1.2 lt.
so volume of water required =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
An example of a reversible reaction is :...The degree of dissociation of pure water at 25°C is found to be 1.8 × 10–9. The dissociation constant, Kd of water, at 2...Assuming no change in volume, calculate the minimum mass of NaCl necessary to dissolve 0.01 mole of AgCl in 100 L soluti...The solubility product of a salt having general formula MX2, in water is: 4 × 10-12. The concentration of M2+ ions ...Sr2+ forms a very unstable complex with NO3−. A solution that was 0.001 M-Sr(ClO4)2 and 0.05 M-KNO3 was found to have on...