Ionic EquilibriumHard
Question
The solubility product of a salt having general formula MX2, in water is: 4 × 10-12. The concentration of M2+ ions in the aqueous solution of the salt is
Options
A.2.0 × 10-6 M
B.1.0 × 10-4 M
C.1.6 × 10-4 M
D.4.0 × 10-10 M
Solution
MX2 ⇋ M+2 + 2X-
S 2S
Ksp = 4s3, S =
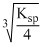 = 1 × 10-4
= 1 × 10-4Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pH of water is 7.0 at 25oC. If water is heated to 70oC, the:...Morphine (C17H19NO3) is administered medically to relieve pain. It is a naturally occurring base or alkaloid. What is th...Amongst the following hydroxides, the one which has the lowest value of Ksp at ordinary temperature (about 25oC) is:...Which of the following will occur if a 0.1 M solution of a weak acid is diluted to 0.01 M at constant temperature?...An amount of 0.10 moles of AgCl(s) is added to one litre of water. Next, the crystals of NaBr are added until 75% of the...