Ionic EquilibriumHard
Question
0.1 millimole of CdSO4 are present in 10 mL acid solution of 0.08 N HCl. Now H2S is passed to precipitate all the Cd2+ ions. The pH of the solution after filtering off precipitate, boiling off H2S and making the solution 100 mL by adding H2O is
Options
A.2
B.4
C.6
D.8
Solution
Cd2+ + H2S → CdS↓ + 2H+
m.moles 0.1
0.2
Total m.moles of H+ in solution after the reaction = 0.2 + 0.8 = 1
∴ [H+] =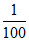 = 0.01 M ⇒ pH = 2.
= 0.01 M ⇒ pH = 2.
m.moles 0.1
0.2
Total m.moles of H+ in solution after the reaction = 0.2 + 0.8 = 1
∴ [H+] =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
An amount of 0.01 moles of solid AgCN is rendered soluble in 1 L by adding just sufficient excess cyanide ion to form Ag...When equal volumes of the following solutions are mixed, precipitation of AgCl (Ksp = 1.8 × 10-1) will occur only w...Three solutions of strong electrolytes, 25 ml of 0.1 M-HX, 25 ml of 0.1 M-H2Y and 50 ml of 0.1 N-Z(OH)2 are mixed, the p...Which of the following solution will have pH close to 1.0 ?...What mass of NaOH should be dissolved in sufficient water to get 20 m3 of an aqueous solution of pH 7.3 at 25o C?...