Ionic EquilibriumHard
Question
pH for the solution of salt undergoing anionic hydrolysis (say CH3COONa) is given by:
Options
A.pH = 1/2 [pKw + pKa + logC]
B.pH = 1/2 [pKw + pKa - logC]
C.pH = 1/2 [pKw + pKb - logC]
D.None of these
Solution
CH3COO- + H2O ⇋ CH3COOH + OH-
∴ [OH-] = C. h = C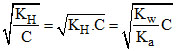
or - log OH = -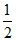 [log Kw + logC - logKa] or pOH =
[log Kw + logC - logKa] or pOH = 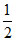 [pKw - log C - pKa]
[pKw - log C - pKa]
Now pH + pOH = pKw ∴ pH =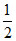 [pKw + log C + pKa]
[pKw + log C + pKa]
∴ [OH-] = C. h = C
or - log OH = -
Now pH + pOH = pKw ∴ pH =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
An ammonia - ammonium chloride buffer has a pH value of 9 with [NH3] = 0.25. What will be the newpH if 500 ml 0.1 M KOH ...At 40o C, the density of heavy water is 1.02 g/ml and its ionic product is 5.1 × 10−15. Which of the following if the on...For the reaction H2(g) + I2 (g) ⇋ 2HI(g) The equilibrium constant Kp changes with :...pH of 0.1M Na2HPO4 and 0.2M NaH2PO4 are respectively : pKa for H3PO4 are 2.2, 7.2 and 12.0....Given Ag(NH3)2+$\rightleftharpoons$ Ag+ + 2NH3, Kc = 7.2 × 10–8 and Ksp of AgCl = 1.8 × 10–10 at 298 K. If ammonia is a...