Ionic EquilibriumHard
Question
An ammonia - ammonium chloride buffer has a pH value of 9 with [NH3] = 0.25. What will be the newpH if 500 ml 0.1 M KOH is added to 200 ml buffer solution (Kb = 2 × 10-5) [log 2 = 0.3]
Options
A.8.4
B.9.6
C.5.6
D.4.4
Solution
5 = pKb + log
[NH4+] = 0.5
[NH3] = 0.25
m moles of KOH added = 50
NH4+ + OH- → NH3 + H2O
100 50 50
50 0 100
pOH = pKb + log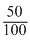
pOH = 4.7 - 0.3 ⇒ 4.4
pH = 9.6

[NH4+] = 0.5
[NH3] = 0.25
m moles of KOH added = 50
NH4+ + OH- → NH3 + H2O
100 50 50
50 0 100
pOH = pKb + log
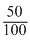
pOH = 4.7 - 0.3 ⇒ 4.4
pH = 9.6
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Pure water is added into the following solutions causing a 10% increase in volume of each. The greatest % change in pH w...The pH of 0.1 M solution of the following compounds increases in the order...In the following reaction HC2O4- (aq) + PO43- (aq) HPO42-(aq) + C2O42-(aq), which are the two Bronsted bases?...The pH value of N/10 NaOH is :...A saturated solution of silver benzoate (AgOCOC6H5) has pH of 8.6. Ka for benzoic acid is 5.0 × 10–5. The value of Ksp f...