Ionic EquilibriumHard
Question
For the reaction H2(g) + I2 (g) ⇋ 2HI(g)
The equilibrium constant Kp changes with :
The equilibrium constant Kp changes with :
Options
A.total pressure
B.catalyst
C.the amounts of H2 and I2 present
D.temperature
Solution
N2(g) + I2(g) ⇋ 2HI(g)2
Kp =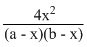
(x = degree of dissoctiation, a & b are initial moles of H2 and I2)
Thus Kp does not depend upon P.
However value of Kp changes with temperature as
2.303 log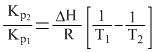
Kp =
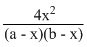
(x = degree of dissoctiation, a & b are initial moles of H2 and I2)
Thus Kp does not depend upon P.
However value of Kp changes with temperature as
2.303 log
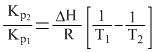
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The molar solubility of Zn(OH)2 in 1 M ammonia solution at room temperature is (Ksp of Zn(OH)2 = 1.6 × 10−17; Kstab of Z...Concentration of the Ag+ ions in a saturated solution of Ag2C2O4 is 2.2 ×10-4 mol L-1 Solubility product of Ag2C2O4...The pH of 0.005 M-NaOH solution is (log 2 = 0.3)...Match list I (Solutions of salts of ...) with list II (pH of the solution is given by) and select the correct answer usi...A particular water sample has 136 ppm CaSO4. What percentage of water by mass must be evaporated in a container before s...