Ionic EquilibriumHard
Question
Which of the following is not correct :
Options
A. [H+] = [OH-] = 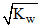 for a neutral solution at all temperatures
for a neutral solution at all temperatures
B.[H+] = [OH-] = 10-7 for a neutral solution at all temperatures
C.[H+] > 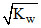 and [OH-] <
and [OH-] < 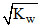 for an acidic solution
for an acidic solution
D.[H+] < 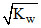 and [OH-] >
and [OH-] > 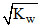 for an alkaline solution
for an alkaline solution
More Ionic Equilibrium Questions
Which is/are correct statements : (i) In any strong acid’s solution, the concentration of [OH-] will be zero. (ii) If DG...A saturated solution of silver benzoate (AgOCOC6H5) has pH of 8.6. Ka for benzoic acid is 5.0 × 10–5. The value of Ksp f...Two buffers, X and Y of pH 4.0 and 6.0, respectively, are prepared from acid HA and the salt NaA. Both the buffers are 0...The solubility of PbCl2 when it is 80% ionized is...At 90o C, the hydronium ion concentration in pure water is 10−6 M. If 100 ml of 0.5 M-NaOH solution is mixed with 250 ml...