Ionic EquilibriumHard
Question
Which is/are correct statements :
(i) In any strong acid’s solution, the concentration of [OH-] will be zero.
(ii) If DGo of a reaction is positive, then the reaction will not proceed at all, in the forward direction for any concentrations of reactants and products.
(iii) Titration curves are drawn for (about the figure shown)
(a) 1M HCl (50 mL) with 1 M NaOH and
(b) 0.01 M HCl (50 mL) with 0.01 M NaOH on the same graph paper they look like:
(i) In any strong acid’s solution, the concentration of [OH-] will be zero.
(ii) If DGo of a reaction is positive, then the reaction will not proceed at all, in the forward direction for any concentrations of reactants and products.
(iii) Titration curves are drawn for (about the figure shown)
(a) 1M HCl (50 mL) with 1 M NaOH and
(b) 0.01 M HCl (50 mL) with 0.01 M NaOH on the same graph paper they look like:
Options
A.i & ii
B.iii only
C.ii only
D.i & iii
Solution
(i) False, [OH-] = 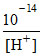 at 25oC
at 25oC
=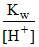 at any other temperature
at any other temperature
(ii) False, ᐃGo = 0 = - RT ln Keq
so, Keq = 1
Hence reaction will proceed significantly in the forward direction.
=
(ii) False, ᐃGo = 0 = - RT ln Keq
so, Keq = 1
Hence reaction will proceed significantly in the forward direction.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The solubility product of CaF2 is 1.08 × 10−10. What mass of CaF2 will dissolve in 500 ml water in order to make a satur...Which of the following is incorrect :-...The precipitate of CaF2 (Ksp = 1.7 × 10-10) is obtained, when equal volumes of the following are mixed :...H3A is a weak tribasic acid with Ka1 = 10−5, Ka2 = 10−9 and Ka3 = 10−13. The value of pX of 0.1 M – H3A solution, where ...The solubility of PbCI2 in water is 0.01 M 25oC. Its maximum concentration in 0.1 M NaCI will be:...