Gaseous StateHard
Question
Consider the following statements:
The coefficient B in the virial equation of state
(i) is independent of temperature
(ii) is equal to zero at boyle temperature PVm = RT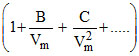
(iii) has the dimension of molar volume
Which of the above statements are correct.
The coefficient B in the virial equation of state
(i) is independent of temperature
(ii) is equal to zero at boyle temperature PVm = RT
(iii) has the dimension of molar volume
Which of the above statements are correct.
Options
A.i and ii
B.i and iii
C.ii and iii
D.i, ii and iii
More Gaseous State Questions
28. Gold has a face centered cubic lattice with an edge length of the unit cube of 407 pm. Assuming the closest packing ...The density of neon will be highest at :...A hypothetical gaseous element having molecular formula Mx may be changed to another gaseous allotrope having molecular ...See the figure-1 : The valves of X and Y are opened simultaneously. The white fumes of NH4Cl will first form at:...Which of the following expression correctly represents the relationship between the average kinetic energy of CO and N2 ...