Gaseous StateHard
Question
A vessel of volume 5 litre contains 1.4 g of nitrogen at a temperature 1800 K. The pressure of the gas if 30% of its molecules are dissociated into atoms at this temperature is :
Options
A.4.05 atm
B.2.025 atm
C.3.84 atm
D.1.92 atm
Solution
N2 → 2N
at t = 0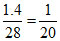 0
0
at t = tf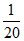 -x 2x
-x 2x
but, x = 30% of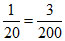
Final number of mole =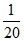 - x + 2x =
- x + 2x = 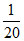 + x =
+ x = 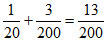
∴ P =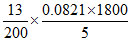 = 1.92 atm.
= 1.92 atm.
at t = 0
at t = tf
but, x = 30% of
Final number of mole =
∴ P =
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
A gas cylinder containing cooking gas can withstand a pressure of 14.9 atmosphere. The pressure guaze of cylinder indica...A and B are two identical vessels. A contains 15 g ethane at 1atm and 298 K. The vessel B contains 75 g of a gas X2 at s...Kinetic theory of gases proves...A mixture of hydrogen and oxygen at one bar pressure contains 20% by weight of hydrogen. Partial pressure of hydrogen wi...X mL of H2 gas effuses through a hole in container in 5 sec. The time taken for the effusion of the same volume of the s...