Gaseous StateHard
Question
At low pressures (For 1 mole), the Vander Waal′s equation is written as
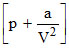 V = RT
V = RT
The compressibility factor is then equal to :
The compressibility factor is then equal to :
Options
A.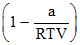
B.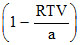
C.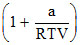
D.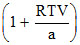
More Gaseous State Questions
Which of the following is not the correct set of pressure and volume at constant temperature and constant moles of gas ?...Henry′s law constants for four gases are given below at 293 K. Which one is more soluble in water at that temperat...The crystal system of a compound with unit cell dimensions ″a = 0.387, b = 0.387 c = 0.504nm and α = β =...Potassium hydroxide solutions are used to absorb CO2. How many litres of CO2 at 1.00 atm and 22oC would be absorbed by a...Which of following statement (s) is true I - Slope of isotherm at critical point is maximum. II - Larger is the value of...