Gaseous StateHard
Question
The average kinetic energy (in joules of) molecules in 8.0 g of methane at 27oC is :
Options
A.6.21 × 10-20 J/molecule
B.6.21 × 10-21 J/molecule
C.6.21 × 10-22 J/molecule
D.3.1 × 10-22 J/molecule
Solution
Average KE = 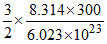 = 6.21 × 10-21 J/molecule.
= 6.21 × 10-21 J/molecule.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
According to the kinetic theory of gases, in an ideal gas, between two successive collisions a gas molecule travels...I, II, III are three isotherms respectively at T1, T2 and T3 as shown in graph. Temperature will be in order...If Z is a compressibility factor, van der Waals equation at low pressure can be written as...20 l of SO2 diffuses through a porous partition in 60 seconds. Volume of O2 diffuse under similar conditions in 30 secon...Equal weights of ethane and hydrogen are mixed in an empty container at 25 oC. The fraction of the total pressure exerte...