Gaseous StateHard
Question
Equal weights of ethane and hydrogen are mixed in an empty container at 25 oC. The fraction of the total pressure exerted by hydrogen is :
Options
A.1 : 2
B.1 : 1
C.1 : 16
D.15 : 16
Solution
Fraction of total pressure exerted by H2 = mole
Fraction of H2 =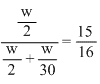
Fraction of H2 =
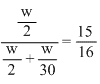
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
The transport of matter in the absence of bulk flow is known as :...A monatomic ideal gas undergoes a process in which the ratio of P to V at any instant is constant and equals to 1. What ...For gaseous state which of the following is incorrect ?...Calculate the radius of He atoms if its Vander Waal′s constant ′b′ is 24 ml mol-1....The crystal system of a compound with unit cell dimensions ″a = 0.387, b = 0.387 c = 0.504nm and α = β =...