JEE Main | 2014Gaseous StateHard
Question
If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Options
A.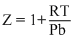
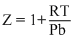
B.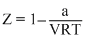
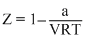
C.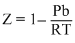
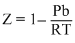
D.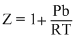
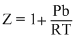
Solution
Compressibility factor (Z) = 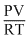
(For one mole of real gas)
van der Waal equation
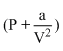 (V - b) = RT
(V - b) = RT
At low pressure
V - b ≈ V
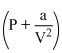 V = RT
V = RT
PV +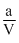 = RT
= RT
PV = RT -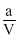
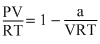
So,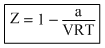
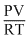
(For one mole of real gas)
van der Waal equation
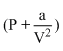 (V - b) = RT
(V - b) = RTAt low pressure
V - b ≈ V
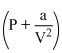 V = RT
V = RTPV +
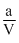 = RT
= RTPV = RT -
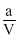
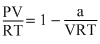
So,
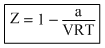
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Helium atom is two times heavier than a hydrogen molecule. At 298 K, the average kinetic energy of a helium atom is...Which one of the following is not a green house gas?...Helium atom is two times heavier than a hydrogen molecule. At 298 K, the average kinetic energy of a helium atom is :...1 mol of a gaseous aliphahatic compound CnH3nOm is completely burnt in an excess of oxygen. The contraction in volume is...An AB2 type structure is found in...