Gaseous StateHard
Question
A compound exists in the gaseous phase both as monomer (A) and dimer (A2). The atomic mass of A is 48 and molecular mass of A2 is 96. In an experiment 96 g of the compound was confined in a vessel of volume 33.6 litre and heated to 273oC. The pressure developed if the compound exists as dimer to the extent of 50 % by weight under these conditions will be :
Options
A.1 atm
B.2 atm
C.1.5 atm
D.4 atm
Solution
Since A and A2 are two states in gaseous phase having their wt ratio 50% i.e. 1 : 1
moles of A =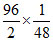 = 1 Moles of A2 =
= 1 Moles of A2 = 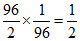
Total mole = 3/2
P = nRT/V.
moles of A =
Total mole = 3/2
P = nRT/V.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Equal masses of H2, O2 and methane have been taken in a container of volume V at temperature 27oC in identical condition...A 40 ml of a mixture of H2 and O2 at 18oC and 1 atm pressure was sparked so that the formation of water was complete. Th...Equal weight of SO2 and O2 are mixed in an container at 25oC. The fraction of the total pressure exerted by O2 is :-...1120.0 mL of ozonized oxygen at S.T.P. weighs 1.76 g. Calculate the volume of oxygen in the ozonized oxygen....The Ca2+ and F- are located in Caf crystal, respectively at face centred cubic lattice points and in...