Gaseous StateHard
Question
1120.0 mL of ozonized oxygen at S.T.P. weighs 1.76 g. Calculate the volume of oxygen in the ozonized oxygen.
Options
A.996 mL
B.896 mL
C.1120 mL
D.784 mL
Solution
n(O2 + O3) = 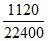 mol = 0.05 mol.
mol = 0.05 mol.
& nO2 + nO3 = n(O2 + O3)⇒ nO2 + nO3 = 0.05 ........ (1)
32nO2 + 48 nO3 = 1.76
⇒ 2nO2 + 3nO3 = 0.11 ........ (2)
From (1) & (2) nO2 = 0.04 mol nO3 = 0.01 mol
Vol of O2 = 22400 ml × 0.04 = 896 ml
& nO2 + nO3 = n(O2 + O3)⇒ nO2 + nO3 = 0.05 ........ (1)
32nO2 + 48 nO3 = 1.76
⇒ 2nO2 + 3nO3 = 0.11 ........ (2)
From (1) & (2) nO2 = 0.04 mol nO3 = 0.01 mol
Vol of O2 = 22400 ml × 0.04 = 896 ml
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
When CO2 under high pressure is released from a fire extinguisher, particles of solid CO2 are formed, despite the low su...Select correct statement(s):...Under what conditions will a pure sample of an ideal gas not only exhibit a pressure of 1 atm but also a concentration o...What volume will be occupied by a sample of gas at 40oC and 720 mm μg if its volume at 20oC and 750 mm μg was ...At 298 K, equal volumes SO2,CH4 and O2 are mixed in empty container. The total pressure of CH4 in mixture is :...