AIPMT | 2014Gaseous StateHard
Question
Equal masses of H2, O2 and methane have been taken in a container of volume V at temperature 27oC in identical conditions. The ratio of the volumes of gases H2 : O2 : methane would be
Options
A.8 : 16 : 1
B.16 : 8 : 1
C.16 : 1 : 2
D.8 : 1 : 2
Solution
Ratio or moles (volume)
⇒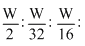
⇒ 16 : 1 : 2
⇒
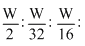
⇒ 16 : 1 : 2
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Which of the following is/are correct for an mono atomic ideal gas molecules :...A monatomic ideal gas undergoes a process in which the ratio of P to V at any instant is constant and equals to 1. What ...The rms velocity of hydrogen is √7 times the rms velocity of nitrogen. If T is the temperature of the gas :...The correction factor 'a' to the ideal gas equation corresponds to...The ratio between the root mean square speed of H2 at 50 K and that of O2 at 800 K is :...