Gaseous StateHard
Question
0.28 g of a gas occupies 224 ml at STP. The gas could be
Options
A.N2
B.CO
C.C2H4
D.C2O4
Solution
ngas = 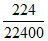 mol = 0.01 mol & M =
mol = 0.01 mol & M = 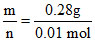 = 28 g/mol
= 28 g/mol
∴ The Gases are N2, CO & C2H4.
∴ The Gases are N2, CO & C2H4.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
A reversible cyclic process for an ideal gas is shown below. Here, P , V and T are pressure , Volume and temperature , r...Under what conditions will a pure sample of an ideal gas not only exhibit a pressure of 1 atm but also a concentration o...1g H2 gas at STP is expanded so that volume is doubled. Hence work done is :-...The given graph represents the variation of Z(compressibility factor ) versus P, for three real gases A, B and C. Identi...Dominance of storage repulsive forces among the molecules of the ges (Z = compressibility factor):...