Gaseous StateHard
Question
At 298 K, equal volumes SO2,CH4 and O2 are mixed in empty container. The total pressure of CH4 in mixture is :
Options
A.0.6 atm.
B.1.2 atm
C.2.4 atm
D.3.6 atm
Solution
Let the wt. of each gas mixed = xg
∴ mole of SO2 =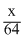
mole of CH4 =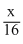
mole of O2 =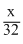
Total number of moles of the three gases
=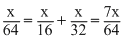
Partical pressure exerted by a gas in the mixture of non-reacting gases (p) is given by
p = × Î¤otal pressure
× Î¤otal pressure
∴ PCH4 =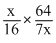 × 2.1 = 1.2 atm
× 2.1 = 1.2 atm
∴ mole of SO2 =
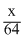
mole of CH4 =
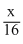
mole of O2 =
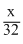
Total number of moles of the three gases
=
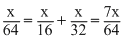
Partical pressure exerted by a gas in the mixture of non-reacting gases (p) is given by
p =
 × Î¤otal pressure
× Î¤otal pressure∴ PCH4 =
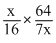 × 2.1 = 1.2 atm
× 2.1 = 1.2 atmCreate a free account to view solution
View Solution FreeMore Gaseous State Questions
Maximum deviation from ideal gas is expected from :...Equal masses of H2, O2 and methane have been taken in a container of volume V at temperature 27oC in identical condition...At Boyle′s temperature, the value of compressibility factor Z = (PVm / RT = Vreal/Videal) has a value of 1, over a...An amount of 1.00 g of a gaseous compound of boron and hydrogen occupies 0.820 liter at 1.00 atm and at 3oC. The compoun...Which of following statement (s) is true I - Slope of isotherm at critical point is maximum. II - Larger is the value of...