Gaseous StateHard
Question
A 0.5 dm3 flask contains gas A and 1 dm3 flask contains gas B at the same temperature. If density of A = 3 g/dm3 and that of B = 1.5 g/dm3 and the molar mass of A = 1/2 of B, the ratio of pressure exerted by gases is :
Options
A.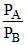 = 2
= 2
B.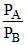 = 1
= 1
C.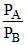 = 4
= 4
D.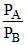 = 3
= 3
More Gaseous State Questions
If Z is the number of atoms in the unit cell that represents the closest packing sequence..... ABC ABC ......, the numbe...The coordination number of a metal crystallizing in a hexagonal close packed structure is :...The compressibility factor of an ideal gas is :...What is the cordination number of sodium in Na2O ?...Van der waal′s equation (V - b) = nRT applicable for :...