Chemical Kinetics and Nuclear ChemistryHard
Question
For The Reaction,
N2O5(g) → 2NO2(g) + 1/2 O2(g)
the value of rate of disappearance of N2O5 is given as 6.25 × 10-3 mol L-1 s-1. The rate of formation of NO2 and O2 is given respectively as -
N2O5(g) → 2NO2(g) + 1/2 O2(g)
the value of rate of disappearance of N2O5 is given as 6.25 × 10-3 mol L-1 s-1. The rate of formation of NO2 and O2 is given respectively as -
Options
A.6.25 × 10-3 mol L-1 s-1 and 6.25 × 10-3 mol L-1 s-1
B.1.25 × 10-2 mol L-1 s-1 and 3.125 × 10-3 mol L-1 s-1
C.6.25 × 10-3 mol L-1 s-1 and 3.125 × 10-3 mol L-1 s-1
D.1.25 × 10-2 mol L-1 s-1 and 3.125 × 10-3 mol L-1 s-1
Solution
Rate of disappearance of reactant = rate of appearance of products
or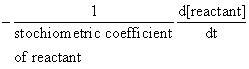
=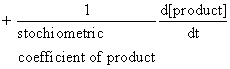
For the reaction,
N2O5(g) → 2NO2(g) + 1/2O2
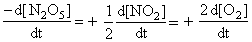
∴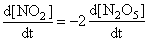
= 2 × 6.25 × 10-3 mol L-1 s-1
= 12.5 × 10-3 mol L-1 s-1
= 1.25 × 10--2 mol L-1 s-1
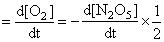
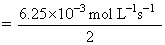
= 3.125 × 10-3 mol L-1 s-1
or
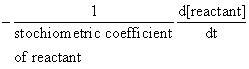
=
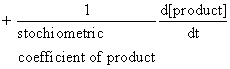
For the reaction,
N2O5(g) → 2NO2(g) + 1/2O2
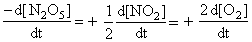
∴
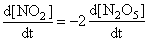
= 2 × 6.25 × 10-3 mol L-1 s-1
= 12.5 × 10-3 mol L-1 s-1
= 1.25 × 10--2 mol L-1 s-1
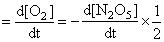
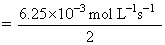
= 3.125 × 10-3 mol L-1 s-1
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Identify compounds readily liberate CO2 gas on heating ?...A certain reaction A → B follows the given concentration (Molarity)-time graph. Which of the following statements ...The correct order of increasing thermal stability of K2CO3, MgCO3, CaCO3 and BeCO3 is :-...Which is not true for a second order reaction ?...Read the following industrial methods for the preparation of H2SO4 and answer the question at the end. Professor Molina ...