Gaseous StateHard
Question
At what temperature root mean square speed of N2 gas is equal to that of propane gas at S.T.P. conditions.
Options
A.173.7oC
B.173.7 K
C.S.T.P.
D.-40oC
Solution
Let Temp (T) where Vrms of N2 = Vrms of C3H8 at STP
=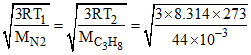
=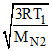 = 393.38
= 393.38
T1 = 173.72 K
=
=
T1 = 173.72 K
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
According to kinetic theory of gases, for a diatomic molecule :...The volume of a gas increases by a factor of 2 while the pressure decreases by a factor of 3. Given that the number of m...At STP the order of mean square velocity of molecules of H2, N2, O2 and HBr is -...A sample of gaseous mixture in the beaker contains 6.08 × 1020 molecules of N2, 0.76 × 1020 molecules of O2, a...The volume temperature graphs of a given mass of an ideal gases constant pressure are shown below. What is the coorect o...