Gaseous StateHard
Question
At STP the order of mean square velocity of molecules of H2, N2, O2 and HBr is -
Options
A.H2 > N2 > O2 > HBr
B.HBr > O2 > N2 > H2
C.HBr > H2 > O2 > N2
D.N2 > O2 > H2 > HBr
Solution
Vrms ∝ 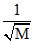 ′M′ is Molecular wt.
′M′ is Molecular wt.
order of M.wt. = H2 < N2 < O2 < HBr
∴ order of Vrms = H2 > N2 > O2 > HBr.
order of M.wt. = H2 < N2 < O2 < HBr
∴ order of Vrms = H2 > N2 > O2 > HBr.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Amongst the following statement, the correct one is:...Which of the following are correct statements ?...For real gases van der Waals equation is written as (V - nb) = nRT where ′a′ and ′b′ are van der...The R.M.S. speed of the molecules of a gas of density 4 kg m-3 and pressure 1.2 ×105 N m- 2 is :...One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0L, 95K) → (4.0 atm, 5.0L, 245K) with a change...