Gaseous StateHard
Question
A sample of gaseous mixture in the beaker contains 6.08 × 1020 molecules of N2, 0.76 × 1020 molecules of O2, and 0.50 × 1020 molecules of CO2. The total pressure is 734 mm of Hg. The partial pressure of O2 would be
Options
A.760.0 mm of Hg
B.76.0 mm of Hg
C.7.6 mm of Hg
D.0.76 mm of Hg
Solution
pO2 = PTotal × χO2 (mole fraction of O2)
χO2 =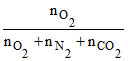
χO2 =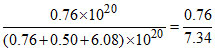
pO2 = PTotal × χO2 = 734 ×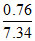
= 76.0 mm of Hg.
χO2 =
χO2 =
pO2 = PTotal × χO2 = 734 ×
= 76.0 mm of Hg.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
The Ca2+ and F- are located in Caf crystal, respectively at face centred cubic lattice points and in...In van der Waals′ equation of state for a non-ideal gas the term that accounts for intermolecular forces is :...The rate of diffusion of SO2, CO2, PCl3 and SO3 are in the following order...If the pressure of a gas contained in a closed vessel is increased by 0.4 % when heated by 1oC its initial temperature m...The given graph represents the variation of Z(compressibility factor ) versus P, for three real gases A, B and C. Identi...