Gaseous StateHard
Question
A gas occupies a volume of 300 cc at 27o Cand 620 mm pressure. The volume of gas at 47o C and 640 mm pressure is :
Options
A.260 cc
B.310 cc
C.390 cc
D.450 cc
More Gaseous State Questions
A bottle of dry ammonia and a bottle of dry hydrogen chloride connected through a long tube are opened simultaneously at...The density of nitrogen is maximum at...A certain volume of argon gas (Mol. Wt. = 40) requires 45 s to effuse through a hole at a certain pressure and temperatu...Kinetic theory of gases proves...When does a gas deviate the most from its ideal behaviour ?...
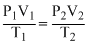
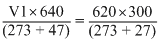
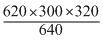 = 310 cc
= 310 cc