Gaseous StateHard
Question
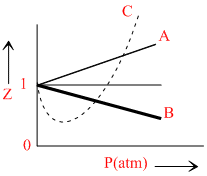
The given graph represents the variation of Z(compressibility factor 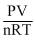 ) versus P, for three real gases A, B and C. Identify the only incorrect statement.
) versus P, for three real gases A, B and C. Identify the only incorrect statement.
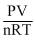 ) versus P, for three real gases A, B and C. Identify the only incorrect statement.
) versus P, for three real gases A, B and C. Identify the only incorrect statement. 

Options
A.For the gas A, a = 0 and its dependence on P is linear at all pressure.
B.For the gas B, b = 0 and its dependence on P is linear at all pressure.
C.For the gas C, which is typical real gas for which neither a nor b = 0. By knowing the minima and the point of intersection, with Z = 1, a and b can be calculated.
D.At high pressure, the slope is positive for all real gases.
More Gaseous State Questions
The temperature of an ideal gas is increased from 120 K to 480 K. If at 120 K the root-mean-square velocity of the gas m...The R.M.S. speed of the molecules of a gas of density 4 kg m-3 and pressure 1.2 ×105 N m- 2 is :...Equal masses of methane and oxygen are mixed in an empty container at 25oC. The fraction of the total pressure exerted b...Three footballs are respectively filled with nitrogen, hydrogen and helium. If the leaking of the gas occurs with time f...A real gas most closely approaches the behaviour of an ideal gas at -...