Chemical Kinetics and Nuclear ChemistryHard
Question
If the rate of reaction, 2SO2(g) + O2(g) 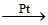 2SO3(g) is given by :
2SO3(g) is given by :
Rate = K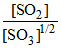
which statements are correct :
Rate = K
which statements are correct :
Options
A.The overall order of reaction is -1/2
B.The overall order of reaction is +1/2
C.The reaction slows down as the product SO3 is build up
D.The rate of reaction does not depend upon concentration of SO3 formed
Solution
Over all order of Rxn = 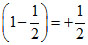
as SO3 for Rate of Rxn slow down.
as SO3 for Rate of Rxn slow down.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
When initial concentration of the reactant is doubled, the half-life period of a zero order reaction...How many coulomb of electricity would be required to reduce the iron in 32.9 g of potassium hexa cyano ferrate (III) K3F...For a second order reaction plots are made for vs time for the reaction, 2A → Product. Pick up the correct sentenc...Consider the following nuclear reactions The number of neutrons in the element L is...As the initial concentration increases from 0.75 to 1.55 M in a reaction, t1/2 decreases from 60 to 29 s. The order of t...