Chemical Kinetics and Nuclear ChemistryHard
Question
T50 (Half-life period) of first-order reaction is 10 minute. Starting with 10 mol L-1, rate after 20 minute is :
Options
A.0.0693 mol L-1 min-1
B.0.0693 × 2.5 mol L-1 min-1
C.0.0693 × 5 mol L-1 min-1
D.0.0693 × 10 mol L-1 min-1
Solution
Conc. after at 2 half life (20 minute) = 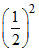 × (Initial conc.)
× (Initial conc.)
=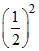 × 10 = 2.5 M.
× 10 = 2.5 M.
Then Rate = k[at] =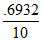 × 2.5 = .06932 × 2.5
× 2.5 = .06932 × 2.5
=
Then Rate = k[at] =
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
If no catalyst (H+) is present in acid hydrolysis of ester (in above question) then rate constant K is :...Upon treatment with ammonical H2S , the metal ion that precipitates as a sulfide is -...For a first order reaction, (A) → products, theconcentration of A changes from 0.1 M to0.025 M in 40 minutes. The ...The initial rate of hydrolysis of methyl acetate (1M) by a weak acid (HA, 1M) is 1/100th of that of a strong acid (HX, 1...During the kinetic study of the reaction, 2A + B → C + D, following results were obtained Based on the above data ...