Chemical Kinetics and Nuclear ChemistryHard
Question
An endothermic reaction with high activation energy for the forward reaction is given by the diagram
Options
A.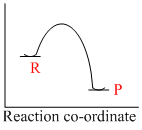

B.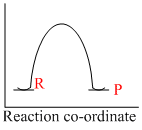

C.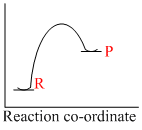

D.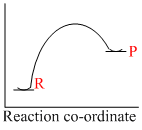

Solution
Reaction which involve absorption of heat energy are called endothermic reactions.Forsuch reaction
∑Hb > ∑HR
In graphs (c) and (d), the heat of product is more than heat of reactants and thus they represent endothermic reaction. But in (d) only small amout of energy is absorbed (less small difference between energy of reactants and products). Thus, (c) represents maximum activation energy.
∑Hb > ∑HR
In graphs (c) and (d), the heat of product is more than heat of reactants and thus they represent endothermic reaction. But in (d) only small amout of energy is absorbed (less small difference between energy of reactants and products). Thus, (c) represents maximum activation energy.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the reaction2N2O5 → 4NO2 + O2The rate of reaction is :...Which of the following nuclear reactions will generate an isotope?...Decrease in atomic number is observed during :...The rate constant of first order reaction is 3 × 10-6 per second. the initial concentration is 0.10 M.The intial ra...Lyophilic colloids are stable due to:...