Chemical Kinetics and Nuclear ChemistryHard
Question
For the reaction A (g) + 2B(g) → C(g) + D(g) 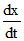 = k [A] [B]2
= k [A] [B]2
Initial pressure of A and B are respectively 0.60 atm and 0.80 atm. At a time when pressure of C is 0.20 atm, rate of the reaction, relative to the initial value is :
Initial pressure of A and B are respectively 0.60 atm and 0.80 atm. At a time when pressure of C is 0.20 atm, rate of the reaction, relative to the initial value is :
Options
A.1/6
B.1/48
C.1/4
D.1/24
Solution
Initial Rate = k [A] [B]2
Rate 1 = k [0.60] [0.80]2 .....(i)
Rate 2 = k [At] [Bt]|2 .....(ii)
Given Rxn
A (g) + 2 B (g) → C (g) + D (g)
0.60 atm 0.80 atm 0 0 t = 0
(0.60 – 0.2) (0.80–0.40) 0.20 0.20 atm
0.40 atm 0.40 atm 0.20 atm 0.20 atm t = t
Put value of pressure of t = t in rate equation (II)
Rate 2 = k [.40] [.40]2
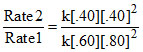
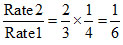
Rate 1 = k [0.60] [0.80]2 .....(i)
Rate 2 = k [At] [Bt]|2 .....(ii)
Given Rxn
A (g) + 2 B (g) → C (g) + D (g)
0.60 atm 0.80 atm 0 0 t = 0
(0.60 – 0.2) (0.80–0.40) 0.20 0.20 atm
0.40 atm 0.40 atm 0.20 atm 0.20 atm t = t
Put value of pressure of t = t in rate equation (II)
Rate 2 = k [.40] [.40]2
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Graph between log k and 1/T (k is rate constant in s-1 and T is the temperature in K) is a straight line. As shown in fi...Which of the following statement is true for the reaction H2 + Br2 → 2HBr. The rate law is = k[H2][Br2]1/2 :...At room temperature, the reaction between NO and O2 to give NO2 is fast, while that between CO and O2 is slow. It is due...For the elementary reaction M → N, the rate of disappearance of M increases by a factor of 8 upon doubling the con...Consider the chemical reaction N2(g) + 3H2(g) → 2NH3(g). The rate of this reaction can be expressed in terms of time der...