Chemical Kinetics and Nuclear ChemistryHard
Question
the density of gold is 19g/cm3. If 1.9 × 10-4g of gold is dispersed in one litre of water to give a sol having spherical gold particles of radius 10 nm, then the number of gold par tiles per mm3 of the so will be
Options
A.1.9 × 1012
B.6.3 × 1014
C.6.3 × 1010
D.2.4 × 106
Solution
Volume of gold present in solution
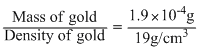
= 0.1 × 10-4 cm3
For spherical particle of gold with radius equal to 10mn The volume of each particle
=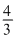 πr3 =
πr3 = 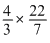 ×(10 × 10-7cm)3
×(10 × 10-7cm)3
=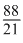 ×
×
Number of gold particle present
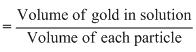
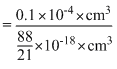
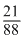 × 1013 particles
× 1013 particles
= 2.4 × 1012 particles
= 2.4 × 1012 particles of gold are present in 1000cm3 (1litre).
∴ Number of particles present per mm3
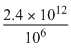 [1L = 106 mm3]
[1L = 106 mm3]
= 2.4 × 106
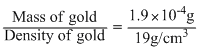
= 0.1 × 10-4 cm3
For spherical particle of gold with radius equal to 10mn The volume of each particle
=
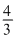 πr3 =
πr3 = 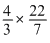 ×(10 × 10-7cm)3
×(10 × 10-7cm)3=
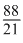 ×
× Number of gold particle present
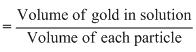
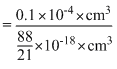
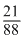 × 1013 particles
× 1013 particles = 2.4 × 1012 particles
= 2.4 × 1012 particles of gold are present in 1000cm3 (1litre).
∴ Number of particles present per mm3
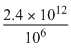 [1L = 106 mm3]
[1L = 106 mm3]= 2.4 × 106
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the non-equilibrium process, A + B → products, the rate is first-order w.r.t. A and second-order w.r.t. B. If ...A catalyst is a substance which...At a certain temperature, the first order rate constant k1 is found to be smaller than the second order rate constant k2...Correct order of rate of SN1 :-...Wrong data for the first order reaction are :-...