Chemical Kinetics and Nuclear ChemistryHard
Question
In a zero-order reaction for every 10o rise of temperature, the rate is doubled. If the temperature is increased from 10oC to 100oC, the rate of the reaction will become:
Options
A.128 times
B.256 times
C.512 times
D.64 times
Solution
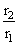 = 2 for every 10o
= 2 for every 10oTemperature is raised from 10o to 100o
So
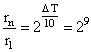
rn = r1 × 512
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Consider a reaction aG + bH → Products. When concentration of both the reactants G and H is doubled, the rate incr...The rate of a first-order reaction is 0.04 mol l-1 s-1 at 10 second and 0.03 mol l-1 s-1 at 20 seconds afterinitiation o...Upon treatment with ammonical H2S , the metal ion that precipitates as a sulfide is -...First order reaction 2N2O(g) → 2N2g + O2(g)has a rate constant of 1.3 × 10-11 S-1 at 270C and 4.5 × 10-1...According to kinetic theory of gases...