Chemical Kinetics and Nuclear ChemistryHard
Question
Which reaction will occur at the fastest rate in water
Options
A.CH3Br + CH3 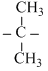 I → CH3 - I + CH3
I → CH3 - I + CH3 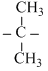 OH
OH
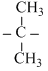 I → CH3 - I + CH3
I → CH3 - I + CH3 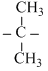 OH
OHB.CH3I + CH3 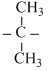 Br → CH3 - Br + CH3
Br → CH3 - Br + CH3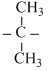 OH
OH
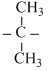 Br → CH3 - Br + CH3
Br → CH3 - Br + CH3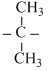 OH
OHC.CH3 - Br + CH3 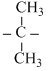 Cl → CH3 - Cl + CH3
Cl → CH3 - Cl + CH3 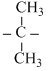 OH
OH
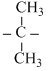 Cl → CH3 - Cl + CH3
Cl → CH3 - Cl + CH3 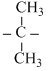 OH
OHD.CH3Cl + CH3 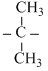 Br → CH3 - Br + CH3
Br → CH3 - Br + CH3 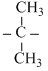 OH
OH
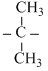 Br → CH3 - Br + CH3
Br → CH3 - Br + CH3 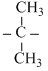 OH
OHSolution
General mechanism will be
In water 3o halide dissociates & shows SN1 mechanism whereas CH3 - X shows (SN2) mechanism.
(CH3)3C - X → (CH3)3C+ + X+
X+ + CH3Y → X - CH3 + Y+
(CH3)3C+ H2O → (CH3)3C - OH
Since I+ is, a good leaving group and a good nucleophile hence reaction ′a′ will be the fastest.
In water 3o halide dissociates & shows SN1 mechanism whereas CH3 - X shows (SN2) mechanism.
(CH3)3C - X → (CH3)3C+ + X+
X+ + CH3Y → X - CH3 + Y+
(CH3)3C+ H2O → (CH3)3C - OH
Since I+ is, a good leaving group and a good nucleophile hence reaction ′a′ will be the fastest.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Which of the following plots in correctly representing the variation in rate of vaporation and rate of condensation with...In the reaction N2 + 3H2 ⇋ 2NH3 = 1 × 102 [N2] [H2]3 - 1 × 10-3 [NH3]2 and at some instant if = 10-5 M2t...A complex reaction 2X + Y → Z, takes place in the following two steps. $X + Y\overset{\quad K_{1}\quad}{\rightarrow}2W$,...The following mechanism has been proposed for the reaction of NO with Br2 to form NOBr: NO(g) + Br2(g) ⇋ NOBr2(g) ...The rate expression for the reaction A(g) + B(g) → C(g) is rate = K·CA2 ·CB1/2. What changes in the initial concentratio...