Chemical Kinetics and Nuclear ChemistryHard
Question
Plots showing the variation of the rate constant (k) with temperature (T) are given below. The plot that follows Arrhenius equation is
Options
A.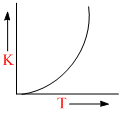

B.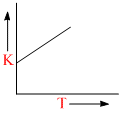

C.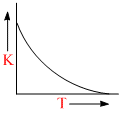

D.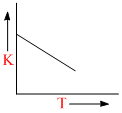

Solution
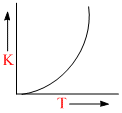
k = Ae-Ea/RT
As T ↑ ; k ↑ exponentially.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Arrange the following in order of decreasing boiling point :-(I) n-butane (II) n-butanol (III) n-butyl chloride (IV) Iso...The rate constant for the reaction, 2N2O5 → 4NO2 + O2 is 3.0 × 10-4s-1. If start made with 1.0 mol L-1 of N2O...The rate constant for the reaction, 2N2O5 → 4NO2 is 3.0 × 10-5s-1 If the rate is 3.40 × 10-5M sec-1, the...Rate constant K = 2.303 min-1 for a particular reaction. The initial concentration of the reaction is 1 mol/litre then r...The half life period of a first order chemical reaction is 6.93 minutes. The time required for the completion of 99% of ...