Chemical Kinetics and Nuclear ChemistryHard
Question
In the following graphical representation for the reaction A → B there are two types of regions :
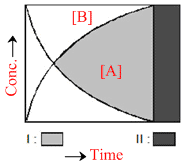

Options
A.I and II both represent kinetic region at different interval
B.I and II both represent equilibrium regions at different time interval
C.I represents kinetic while II represents equilibrium region
D.I represents equilibrium while II represents kinetic region
Solution
I represent kinetic but II represent equilibrium region
Because at equilibrium the concentration remains constant.
Because at equilibrium the concentration remains constant.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The initial rate of hydrolysis of methyl acetate (1M) by a weak acid (HA, 1M) is 1/100th of that of a strong acid (HX, 1...The decomposition of H2O2 can be followed by titration with KMnO4 and is found to be a first order reaction. The rate co...If r = k [A]02 [B]0 for a reaction, by what factor is the initial rate multiplied if the [A]0 is multiplied by 1.5 and t...In general, the rate of a reaction can be increased by all the factors except...In a zero-order reaction for every 10o rise of temperature, the rate is doubled. If the temperature is increased from 10...