Chemical Kinetics and Nuclear ChemistryHard
Question
The decomposition of H2O2 can be followed by titration with KMnO4 and is found to be a first order reaction. The rate constant is 4.5× 10-2. In an experiment, the initial titer value
Options
A.4.5 × 10-2 × 5 min
B.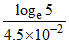 min
min
C.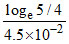 min
min
D.None of the above
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Figure shows human urinary system with structures labelled A to D. Select option which correctly identifies them and giv...For the elementary reaction M → N, the rate of disappearance of M increases by a factor of 8 upon doubling the con...In a reaction, A + B → Product, rate is doubled when the concentration of B is doubled, and rate increases by a fa...Consider the reaction : Cl2(aq) + H2S(aq) → S(s) + 2H+(aq) + 2Cl- (aq) The rate equation for this reaction is rate...Units of rate constant of first and zero order reactions in terms of molarity M unit are respectively...