ElectrochemistryHard
Question
Pure water is saturated with pure solid AgCl, a silver electrode is placed in the solution and the potential is measured against normal calomel electrode at 25oC. This experiment is then repeated with a saturated solution of AgI. If the difference in potential in the two cases is 0.177V. What is the ratio of solubilty product (solubilty) of AgCl and AgI at the temperature of the experiment ?
Options
A.103
B.106
C.102
D.102
Solution
Ag → Ag+ + e-
E1 = Eoxid + Ecalomel
= E′ -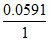 log + Ecalomel
log + Ecalomel
E2 = E′ -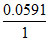 log + Ecalomel
log + Ecalomel
E2 - E1 = 0.177 = 0.0591 log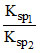
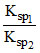 = 103.
= 103.
E1 = Eoxid + Ecalomel
= E′ -
E2 = E′ -
E2 - E1 = 0.177 = 0.0591 log
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
When a lead storage battery is charged...At 25°C, the solubility product of CuCl is 2.0 × 10−7 and $E_{Cl^{-}|CuCl|Cu}^{o}$ is 0.128 V. The value of $E_{Cu^{+}|C...Which one of the following statements is incorrect regarding an electrochemical cell?...ᐃG is the available energy (energy produced) during the electrochemical reaction in galvanic cell which can be con...The conductivity of a saturated solution of AgCl at 298 K was found to be 3.40 × 10−6 Ω−1 cm−1; the conductivity of wate...