ElectrochemistryHard
Question
Na-amalgam is prepared by electrolysis of NaCl solution using liquid Hg as cathode. How long should the current of 10 amp. is passed to produce 10% Na - Hg on a cathode of 10 gm Hg. (atomic mass of Na = 23).
Options
A.7.77 min
B.9.44 min.
C.5.24 min.
D.11.39 min.
Solution
90 gm Hg has 10 gm Na
∴ 10 gm Hg =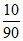 × 10 =
× 10 = 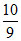 gm Na
gm Na
∴ weight of Na =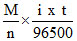
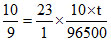 [∴ Na+ + e → Na]
[∴ Na+ + e → Na]
∴t =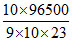 = 7.77 min
= 7.77 min
∴ 10 gm Hg =
∴ weight of Na =
∴t =
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A hydrogen electrode placed in a buffer solution of CH3COONa and CH3COOH in the ratios of x : y and y : x has electrode ...The chemical reaction,2AgCl(s) + H2(g) → 2HCl(aq) = 2Ag(s)Taking place in a galvanic cell is represented by the no...What is the amount of chlorine evolved, when 2 amp of current is passed for 30 minutes in an aqueous solution of NaCl?...The Gibbs energy for the decomposition of Al2O3 at 500oC is as follows : Al2O3 → Al + O2, ᐃrG = + 966 kJ mol...The cell potential for the voltaic cell depicted below is 0.109V under standard conditions ; Pb(s)| Pb2+(aq.)||Ni2+(aq.)...